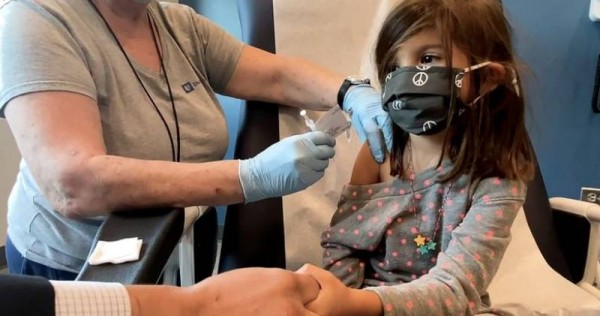
Pfizer Inc and BioNTech have asked US regulators to approve emergency use of their Covid-19 vaccine for children aged five to 11, Pfizer said in a tweet on Thursday (Oct 7).
The US Food and Drug Administration has set a date of Oct 26 for its panel of outside advisers to meet and discuss the application, making it possible for kids to begin receiving the vaccines soon afterwards.
A rapid authorisation of the vaccine in young kids could help mitigate a potential surge of cases this fall, with schools already open nationwide.
The vaccine could be ready for roll-out as early as November pending approval from federal regulatory health agencies, White House Covid-19 response coordinator Jeffrey Zients said on Thursday.
Once the authorisation is complete, Zients said: “We are ready. We have the supply. We’re working with states to set up convenient locations for parents and kids to get vaccinated including paediatricians’ offices and community sites.”
Children currently make up about 27 per cent of all US coronavirus cases and an increasing percentage of hospitalisation, according to the American Academy of Pediatrics.
That reflects the high contagiousness of the Delta variant among unvaccinated people.
While kids are less susceptible to severe Covid-19, they can spread the virus to others, including vulnerable populations that are more at risk of extreme illness.
The vaccine is already authorised in teens aged 12 to 15 and fully approved for ages 16 and up, and has been shown to induce a strong immune response in the target age group in a 2,268 participant clinical trial, the companies said on Sept 20.
The two drugmakers are also testing the vaccine in children ages two to five and children aged six months to two years, with data expected in the fourth quarter.
This article was first published in Asia One . All contents and images are copyright to their respective owners and sources.